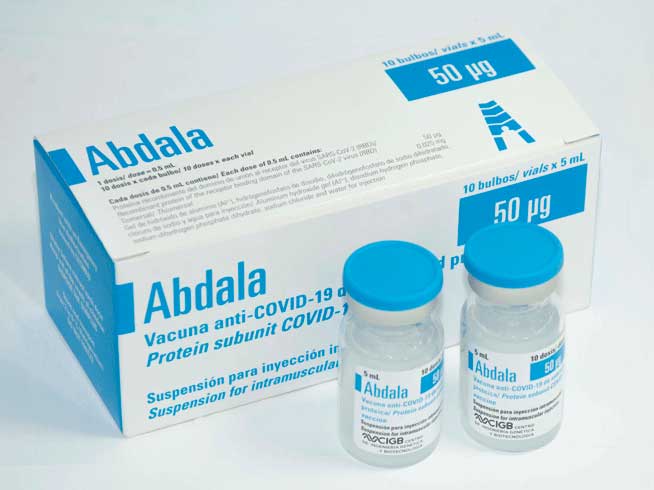
 Researchers at the Center for Genetic Engineering and Biotechnology (CIGB) plan to submit the dossier of the Cuban vaccine Abdala to the World Health Organization (WHO) in March, as part of the process to obtain its authorization for emergency use.
Researchers at the Center for Genetic Engineering and Biotechnology (CIGB) plan to submit the dossier of the Cuban vaccine Abdala to the World Health Organization (WHO) in March, as part of the process to obtain its authorization for emergency use.
Havana, Cuba.- Miladys Limonta Fernández, the coordinator of anti-COVID-19 vaccine candidate development projects of the CIGB, told the Cuban News Agency that the authorization process includes sending a letter with the expression of interest, an initial meeting to present the general information on the vaccine, and then the dossier is delivered.
She said that the letter of interest has already been sent, and they are currently working on completing the dossier, which is made up of five modules with information on the producers, summary of experts, quality data, stability, production of the active pharmaceutical ingredient and the finished product, production scales, pharmacology and toxicology studies in animals, and clinical trials.
Once the dossier has been reviewed, the WHO experts can inspect the production plants, which in the case of Abdala would be at the CIGB-Mariel Biotechnological Industrial Complex, Limonta Fernández said.
The expert explained that the CIGB has experience in the prequalification of vaccines by the international health organization, with Heberbiovac HB, against hepatitis B, and Quimi-Hib, a conjugated vaccine against meningitis, both produced by the institution.
Abdala, the first anti-COVID-19 vaccine developed, produced, and authorized for emergency use in Latin America. (RHC)